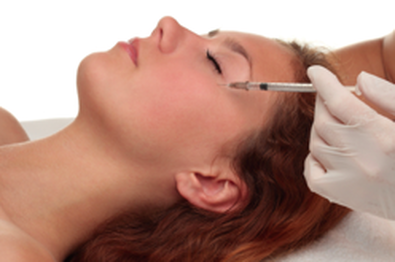
Botulinum toxin has a history goes back almost two hundred years.
In 1822, a German doctor named Justinius Kerner, suggested that botulinum toxin injections might be used in the treatment of excessive sweating or hyperhidrosis. Khalaf Bushara and David Park were the first to show that botulinum toxin injections inhibit sweating (axillary hyperhidrosis). This was the first demonstration of non- muscular use of botulinum toxin. The sweat glands no longer receive the signal to produce sweat. Botox® is now regularly used for this condition.
Researchers discovered in the 1950s that injecting overactive muscles with minute quantities of botulinum toxin type-A would result in decreased muscle activity by blocking the release of acetylcholine from the neuron by preventing the vesicle where the acetylcholine is stored from binding to the membrane where the neurotransmitter can be released. In the absence of signals from the nerves, the muscles aren’t able to contract leading to a softening of the wrinkles. This will effectively weaken the muscle for a period of three to four months.
In the late 1960s, Botox® was used in American clinics for the treatment of strabismus (crossed eyes) by Alan Scott.
Twenty years later, the real breakthrough came in the wider application of Botox® when Allergan, the American manufacturer of Botox®, was granted approval to market the drug by the Federal Drugs Agency. This gave Allergan the license to sell its new product throughout the U.S. Since then, Botox® has been registered and used in over 90 countries around the world.
Other versions of botulinum toxin are Botox®, Botox Cosmetic® & Vistabel® Dysport® / Azzalure®, Xeomin® / Bocouture® (all type A varieties) and Neurobloc® (type B). In the near future there may be 2 new botulinum type A varieties to the market PurTox® and RT001 from Revance® Therapeutics.
The cosmetic effect of BTX-A on wrinkles was originally documented by a plastic surgeon from Sacramento California Dr. Richard Clark.
In 1987 via a Canadian Ophthalmologist, Dr. Jean Carruthers. had been treating her patients who suffered from blepharospasm with Botox®. Dr. Carruthers observed that a "side-effect" of this treatment was the reduction of crow's feet and wrinkles around the eyes. She then decided to test these findings scientifically together with her husband a dermatologist.
Since then an enormous amount of clinical trial work and patient experience has been established using Botox® for the treatment of wrinkles. Dynamic wrinkles are caused by repeated muscle contraction or facial expressions. Some of these wrinkles, like worry lines, frown lines or laugh lines, are often named after facial expressions. When we’re young, our skin is more elastic and the lines easily bounce back to a normal, wrinkle free look. But as we age, these lines become more permanent fixtures in our faces.
If the wrinkle is not caused by muscular contractions, you might consider another treatment option like dermal facial fillers.
There are three major types of dynamic wrinkles that can be very effectively treated with Botox®.
• Glabellar lines (frown lines)
• Forehead furrows (worry lines)
• Crow’s feet, wrinkles radiating out from the corners of the eyes
In 2010 The American Society for Aesthetic Plastic Surgery rated treatment with botulinum toxin the most popular aesthetic procedure in the U.S.
Science has discovered many other clinical uses for botulinum toxin including: wound healing, spasticity in adults and children, strabismus, blepharospasm, incontinence, back pain, achalasia, migraine, dystonia, anal fissure, vocal cord dysfunction, vaginismus, which have all been treated successfully with this important product.
There are currently different types of botulinum toxin commercially available: varieties of type A toxin & type B toxin, from which one is for topical use
Botox®, Botox Cosmetic® & Vistabel®
Botox® was the first botulinum toxin to gain a cosmetic licence endorsing its use for glabellar lines and wrinkles marketed also under the brand names Vistabel® and Botox Cosmeticâ
Botox® is made from purified type - A neurotoxin and manufactured and marketed by Allergan, Inc.
www.botoxcosmetic.com
Dysport® & Azzalure®
Azzalure®, Clostridium botulinum toxin (type) A haemagglutinin neurotoxin complex / purified type - A neurotoxin, has been available as the brand name Dysport®. It gained approval for cosmetic use and is marketed for this indication under the brand name Azzalure®. It is manufactured by the French company Ipsen and marketed by Galderma.
www.dysport.com
Xeomin® or Bocouture® in cosmetic use
Clostridium botulinum type - A neurotoxin complex. Also known as NT-201, botulinum neurotoxin free from complexing proteins. Manufactured by Merz.
www.bocouture.com
Myobloc® or Neurobloc® contains botulinum toxin type B and is commercialized by Solstice Neurosciences.
Lanzhou Institute (China) manufactures a BTX-A product.
Neuronox®, a BTX-A product, was introduced by Medy-Tox Inc. of South Korea, in 2009.
PurTox®, a BTX-A product is currently in Phase III Mentor Corporation
PurTox
Linurase® by Prollenium Medical Technologies
Revance® therapeutics has a new BTX-A currently in Phase II
http://www.revance.com/products.html
Please be aware that practitioners can use all brands available, though not all products are granted the same licensed indications. Practitioners should inform you if they are using the product outside of their current licensed indications.
The Leading Injectable Centers of the World® Quality in Beauty



 RSS Feed
RSS Feed